- Stocks surge at DSE, CSE for second straight day |
- REHAB seeks Tk 3000 crore special fund support housing sector |
- Trump, Putin discuss Iran conflict, Ukraine settlement in phone call |
- US Launches ‘Most Intense’ Strikes as Iran Vows Response |
- Family Card Scheme to Cover 4 Crore Families in 5 Years |
Antibiotic Resistance: A Quiet Health Emergency in BD
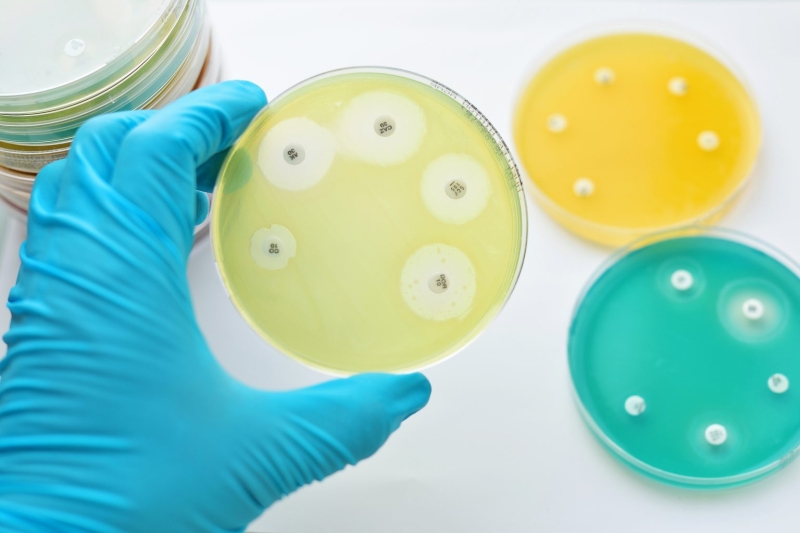
Antibiotic resistance in Bangladesh has quietly escalated into a severe public health challenge, with scientists, doctors, and global health agencies warning that the country is at risk of losing the effectiveness of medicines that once cured common infections. This issue is part of a wider global crisis identified by the World Health Organization (WHO) as one of the top 10 threats to human health worldwide.
At its core, antibiotic resistance happens when bacteria change in ways that reduce or eliminate the effects of antibiotics designed to kill them. That can make once‑treatable illnesses much harder or even impossible to cure, putting patients at risk of prolonged sickness, higher healthcare costs, and death.
In Bangladesh, recent global health estimates put the number of deaths attributable to antibiotic-resistant infections at roughly 23,500 in 2021 alone, with nearly 97,000 deaths associated with resistant bacteria that year.
Despite this, awareness in the general population remains limited. A 2023 study in Bangladesh found gaps in public knowledge about antibiotics and resistance, with many people unsure how resistance develops or why completing a full course of antibiotics matters.
The WHO first declared antimicrobial resistance (AMR) a major global health danger more than a decade ago, placing it among the most urgent crises facing humanity. By 2019, resistant infections were directly linked to 1.27 million deaths and contributed to nearly 5 million deaths globally. Without action, projections suggest that deaths attributable to AMR could climb into the tens of millions annually by 2050.
This global trend reflects regional struggles. In South Asia, including Bangladesh, up to one in three bacterial infections are now resistant to common treatments, according to the latest WHO surveillance data.
Research from hospitals and diagnostic centres shows alarming patterns of resistance among common bacterial pathogens in Bangladesh. Studies conducted in Dhaka and other regions reveal high rates of resistance in multiple bacterial species against widely used antibiotics:
· Escherichia coli, a common cause of urinary and bloodstream infections, shows significant resistance to key antibiotics.
· Klebsiella species and Pseudomonas aeruginosa display reduced susceptibility to drugs often used as first‑line treatment.
· Staphylococcus aureus, including methicillin‑resistant strains (MRSA), continues to challenge clinical care with resistance to commonly used antibiotics.
· Resistance in Gram‑positive and Gram‑negative bacteria has been observed across urine, wound, and other clinical samples, often leaving limited drug options for treatment.
One nationwide cross‑sectional study reported that E. coli was the most common resistant pathogen (32.5 % of isolates), with high levels of resistance to third‑generation cephalosporins and a large proportion of isolates showing resistance to β‑lactam antibiotics.
Research at Bangladesh Medical University (BMU) also found that more than 21 % of patients in specialised units showed resistance to antibiotics, with more than half of intensive care unit (ICU) patients exhibiting multi‑drug resistance.
Surveillance data suggests that a substantial number of patient samples collected were already taken after antibiotic treatment had begun, underlining how routine therapy may be contributing to resistance development.
Doctors and public health experts are clear that the rampant misuse and overuse of antibiotics are at the heart of the problem in Bangladesh. Unlike many high‑income countries where prescriptions are tightly regulated, antibiotics in Bangladesh are widely available without a doctor’s oversight. Patients commonly self‑medicate for fevers, coughs, or stomach issues with antibiotic drugs purchased over the counter, often without completing recommended courses.
Prescriptions written by under‑qualified practitioners and aggressive marketing by pharmaceutical companies further fuel inappropriate antibiotic use. In many rural and urban drug shops, antibiotics are dispensed freely, encouraging consumers to rely on them as first‑line treatments even when unnecessary.
Agricultural practices also play a major role. Farmers routinely add antibiotics to animal feed and poultry water to promote growth and prevent disease, a practice that accelerates the spread of resistant bacteria from animals to humans through food chains and the environment.
Bangladesh’s healthcare infrastructure struggles to keep pace with the growing threat. Infection prevention and control measures are often inadequate in hospital settings. Poor sanitation, insufficient clean water supplies, and irregular infection control practices create environments where resistant organisms thrive and spread.
Diagnostic laboratories are spread thin, and many cannot accurately identify resistant strains in clinical samples due to limited infrastructure or resources. Without effective laboratory support, doctors must sometimes prescribe broad‑spectrum antibiotics empirically, which further accelerates resistance development.
Antibiotic stewardship programmes that would guide clinicians on rational antibiotic use are rare in most hospitals and clinics, limiting opportunities for front‑line intervention to curb the crisis.
Bangladesh has taken policy steps to address AMR, including a National Strategy and Action Plan for Antimicrobial Resistance Containment (2021–2026), which emphasises a One Health approach involving human, animal, and environmental health sectors. This plan aligns with global frameworks championed by the WHO and other international partners.
However, implementation has lagged. Regulatory enforcement to restrict over‑the‑counter antibiotic sales remains weak. Surveillance systems for tracking antibiotic use and resistance patterns exist, but their coverage and standardisation need significant improvement to inform effective responses.
Healthcare professionals warn that if antibiotic resistance continues to grow unchecked in Bangladesh, routine infections could become deadly. Minor wounds, urinary tract infections, and post‑operative complications that once responded easily to standard antibiotics could become life‑threatening.
Projections from global research suggest that without urgent action, millions of lives could be lost to resistant infections annually by mid‑century, with disproportionate impacts in low‑ and middle‑income countries such as Bangladesh.
Experts emphasise that addressing antibiotic resistance in Bangladesh will require coordinated effort across multiple fronts:
Laws restricting the sale of antibiotics without a prescription must be enforced at pharmacies and drug shops nationwide.
Standardised, nationwide systems to monitor resistant infections and antibiotic usage are critical. This requires investment in labs, training for microbiologists, and data systems to track trends.
Awareness initiatives must reach all corners of the country to teach people why antibiotics must be used responsibly and why completing prescribed courses matters.
Hospitals and clinics must adopt antibiotic stewardship programmes that guide clinicians on appropriate use and reduce unnecessary prescriptions.
Coordination between human health, animal agriculture, and environmental sectors is essential to reduce antibiotic misuse in farming and limit the spread of resistant bacteria across ecosystems.
Antibiotic resistance in Bangladesh has grown from a hidden health issue into a looming crisis. The convergence of widespread misuse, weak regulation, limited diagnostics, and low public awareness has created fertile ground for resistant bacteria to flourish.
Experts insist that change is possible. With strengthened policies, improved surveillance, and sustained public engagement, Bangladesh can slow the advance of resistance and protect future generations from the silent epidemic now unfolding.

